Chemo-rated gloves protect your healthcare workers exposed to hazardous drugs such as chemotherapy preparations as well as being a key consumable to help you achieve USP800 compliance. But selecting the right chemical protective glove is not as straightforward as it may seem.
Chemotherapeutic chemicals are either:
- cytostatic – destroy cells
- cytotoxic – inhibit the growth or multiplication of cancer cells.
So, by nature, these drugs are hazardous and can cause damage to bare skin and other exposure that is not intended. ASTM gloves are one part of an overall strategy of personal protective clothing that provide an effective barrier between workers and hazardous drugs…protecting the worker and your product.
Let’s explore some of the points to consider when selecting chemo grade gloves for your pharma manufacturing cleanroom, hospital lab, or other healthcare facility.
Chemo-Approved Glove Materials
Below are the most common cleanroom glove materials and the list of substances they are generally resistant to:
- Latex or other natural rubber – water, alcohol, and some ketones
- Neoprene – water, alcohol, detergent, acid and other caustics, and ketones
- Vinyl (PVC) – water, alcohol, detergent, weak acid and other weak caustics, and ketones
- Nitrile – Oil, fuel, some organic solvents, and weak acid and other weak caustics
- Butyl rubber – Acid and other caustics, some ketones, and some esters
- Polyvinyl alcohol – aliphatic, aromatic, and halogenated hydrocarbons; some ketones, and some esters
- Viton (a type of rubber) – aromatic and halogenated solvents
- Polyethylene and ethylene vinyl alcohol (plastic film) – broad categories of chemical classes
Source: AIHA Chemical Protective Clothing, 2nd edition.

Chemical Resistance Testing
The three main types of chemical resistance testing are discussed below.
Chemical Degradation Testing
Degradation refers to the change in a product’s physical properties after coming into contact with a chemical hazard such as chemotherapy. Testing is conducted by putting the glove in contact with a specific chemical at a pre-determined time and temperature followed by an assessment of physical changes including discoloration, deformation, thinning, shrinkage, tensile strength, and other properties. Changes in weight and surface area can be used to rate degradation.
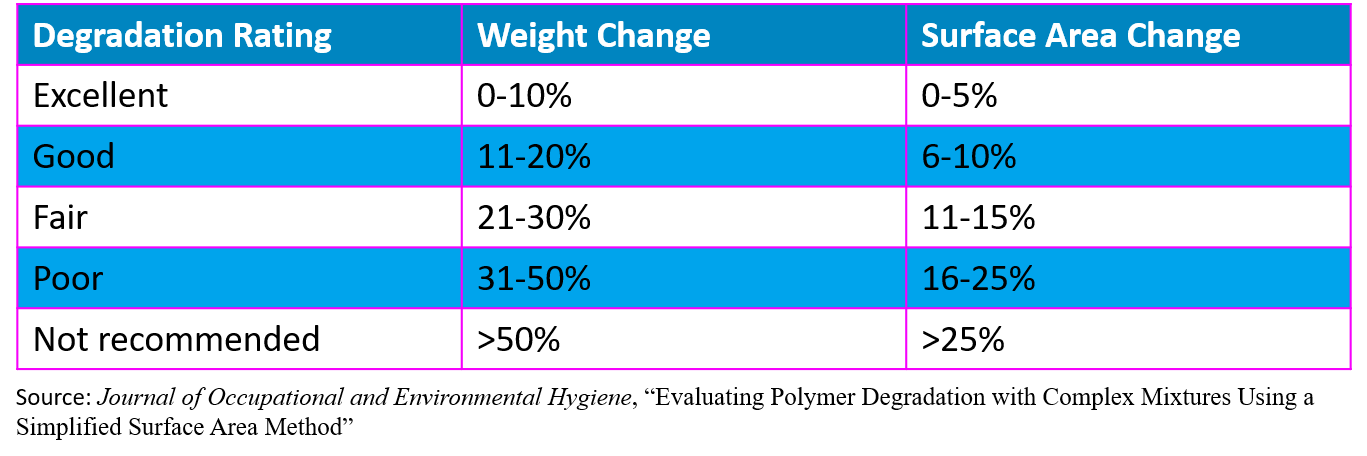
Common degradation testing standards include ASTM D471 and EN 374-4.
Selecting a chemo-approved glove that is not affected by exposure to chemotherapy preparations is vital to protection of workers and your products.
Penetration Testing
This type of chemical resistance testing targets determination of flow of a liquid or gas through the glove’s surface. Penetration could come from small holes of the material or from defects. Chemical, water, or air leak testing, or a combination, can be used to assess penetration.
Glove penetration standards include:
- American Society for Testing and Materials (ASTM) Methods F903 and D5151
- U.S. Food and Drug Administration (FDA) method for medical exam gloves (21 CFR 800.20)
- European Union Method 374-2.
These standards determine the number of defects detected per 100 gloves that is often referred to as the acceptable quality level (AQL) which is a numeric grade ranging from 0.65 to 4.0.
Selecting a chemo-tested glove with a low probability of a defect or material construction that enables penetration is critical to protect against chemotherapy and other hazardous drug exposure.
Chemical Permeation Testing
Permeation testing evaluates the molecular movement of a hazard such as chemotherapy solutions through the glove material.
Tests assess breakthrough time (BT) and permeation rate (PR):
- The BT is duration of time, in minutes, that passes prior to first detection of the chemical passing through the glove material.
- The PR is either the maximum rate or steady-state rate of permeation in units of microgram per square centimeter per minute (µg/cm2/min).
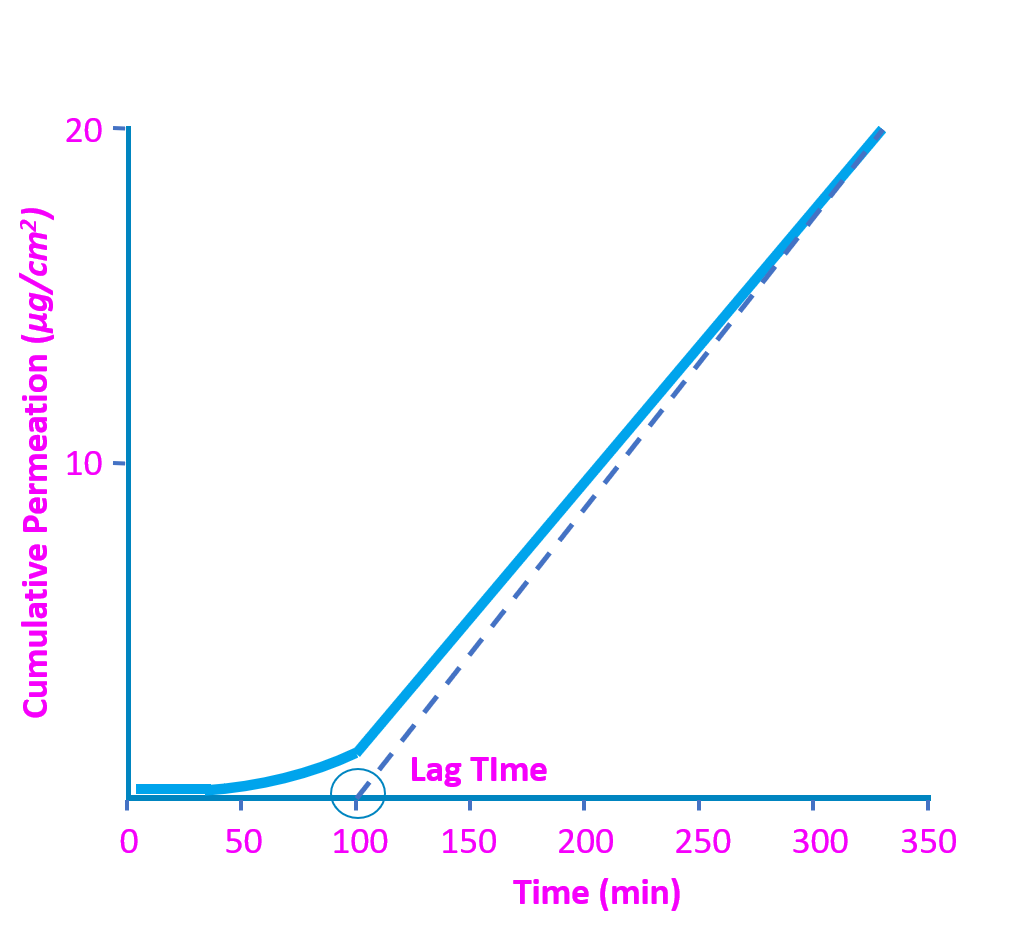
These 2 data points, BT and PR, together express how quickly the chemical can permeate through the glove.
Common testing standards include ASTM F739 and EN 374-3.
Selecting a chemo-grade glove with a high enough BT and lower PR is key to ensuring worker safety and product purity.
Key Considerations for Choosing a Chemo Glove
- Select gloves with a lower AQL so that there is a lower probability of penetration of HDs.
- Avoid gloves that degrade in the presence of chemicals that are used in your processes.
- Select products with permeation rankings that include BT and PR.
- Balance thickness with dexterity, higher thickness is more protective but may affect comfort and tactile sensitivity.
Free Advice on Choosing a Chemo Glove
Our BioClean partners have a handy cleanroom glove selection tree diagram that helps you select gloves that suit your application. Plus the chart increases your understanding of the glove attributes that are important in your environment.
Or give our cleanroom consultants a call. They can answer all your questions about the best gloves for your pharmaceutical cleanroom or other healthcare facility. Our direct relationships with top quality US supplies of chemo-rated gloves assure that we are informed on the latest chemo/HD gloves and their testing requirements.
We can help you be confident that your gloves match your needs. We offer free cleanroom glove samples so you can try before you buy at the lowest prices guaranteed.
We can also assist you with USP800 compliance procedures such as using two pairs of disposable single-use gloves to effectively protect against chemotherapy exposure.