This pharmacy cleanroom cleaning guide is an overview of the cleanroom chemicals, products, and tools and how they are typically used for compounding pharmacy applications. Each cleanroom and its applications have unique cleaning requirements. At no time should this guidance replace existing documents and procedures established by the pharmacy facility unless written permission is obtained from the facility manager. Consult your cleanroom standard operating procedures for protocols specific to your critical environment.
Cleaning and sanitizing ISO Class 5 (Class 100) cleanrooms such as compounding pharmacies and hospital pharmacies to meet USP Chapter 797 requirements requires consistently utilized protocols. While some USP requirements mean large investments of capital, cleanroom cleaning protocols can be implemented at a relatively low cost. However, proper apparel use and cleaning routines do require staff cooperation that is most easily earned through education and clear written procedures.

An understanding of how proper garbing and cleaning can provide immediate and substantial risk reduction is the key to adopting the needed changes.
This guideline is an overview of the cleaning and disinfection guidelines with a brief overview of monitoring cleanliness and sterility for cleanest ISO Class 5 to less-strict 8 cleanrooms. Federal standards mandate the number of particles equal to and greater than 0.5mm measured in one cubic foot of air for each class of cleanroom.

Establishing a Cleanroom Cleaning Routine
When a pharmacy cleanroom has implemented primary and secondary engineering controls and realized consistent use of the proper PPE and particulate-control measures, the weak link of cleaning procedures can break the chain of sterility and cleanliness. Lapses in cleaning protocols result in viable particles, those of a biological nature, to void asepsis or allow infiltration of non-viable particles that while not biologically active, serve as transportation for micro-organisms.
For some pharmacy cleanrooms, a professional cleanroom cleaning service can assist with keeping environmental hygiene at its highest. But in many pharmacies, the staff cleans the controlled environment. In hospitals, the environmental services department may have this responsibility at least partially as some hospital pharmacies utilize their environmental services department into weekly or monthly cleaning cycles, while the cleanroom staff complete daily cleaning protocols.
The most important aspect is not who cleans the pharmacy, but rather what they know. Detailed cleaning protocol training is vital. Download a high-level sample of an ISO5 compounding cleanroom cleaning routine. Periodic supervision of the cleaning in progress as well as record-keeping of the cleaning routine are required to ensure the cleaning is performed as planned and documented for compliance with USP requirements.
Cleanroom Cleaning Protocols for Special Situations
When bringing a cleanroom online for the first time or if contamination is found, especially with tough sporicides, your designed cleaning protocol can be performed multiple times to achieve the level of cleanliness and sterility needed for your ISO 5 pharmacy.
- Triple clean – sanitizing the critical area and anteroom 3 times. First cleaning with a detergent-based cleaner followed by 3 disinfecting cycles: complete cleaning with 2 different disinfectants then a sporicidal agent
- 9-time clean – basically doing the triple clean 3 times which is an investment no pharmacy wants to make, but for worst case scenarios, it is sometimes required.
What to Use to Clean Your Pharmacy Cleanroom
Cleaning and sanitizing agents are critical to the process. Cleaning and disinfecting are two separate actions that must be performed in a clean then disinfect order to thoroughly clean a facility.
Cleaning
Cleaning using a detergent or surfactant solution is for removing dirt, grime, grease, and particulates and for removing microbial contamination from a surface. Disinfection is the application of a chemical with germicidal properties to eliminate micro-organisms.
Cleaning comes before disinfection. It is a bit like cleaning at home. If you do not remove the crumbs and spills from the kitchen counter with a wet paper towel, then disinfecting with wipe with a bleach-based spray is less effective and takes more product to ensure sanitization. It is the same in a cleanroom. For example, use a dry microfiber mop or sticky roller to remove debris from the floor, the most contaminated part of your environment given that you walk on it and that air circulation is designed to force contaminants down. Then, after cleaning the floor, apply disinfectant thoroughly with a mop.
Disinfecting
Sanitization is vital to a USP 797-compliant compounding pharmacy or hospital pharmacy. But it is one of the areas that can be very confusing when designing your cleaning protocols. For one thing, there are many types of disinfectants. They can be grouped into two general categories: oxidizing and non-oxidizing agents. Each category kills different types organisms: oxidizing agents are broad spectrum with the power to kill spores, the toughest of micro-organisms to eradicate while non-oxidizing agents are organism specific. Note that the more general oxidizing agents are non-specific creating risks for humans cleaning so if they are part of your protocol, use greater caution to protect cleaning workers.
Non-oxidizing agents include:
- Alcohols
- Quaternary Ammonium Compounds (QAC or quats)
- Phenolics (the FDA notes that phenolics are not preferred and they are banned for use in Canada).
Oxidizing agents include:
- Halogens
- Peracetic Acid
- Hydrogen Peroxide.
Isopropyl alcohol (IPA) is commonly used as a sanitizing agent for surfaces in ISO Class 5 compounding cleanrooms, but 3% hydrogen peroxide and 2% sodium hypochlorite (bleach) solutions also effectively sanitize. The Center for Disease Control (CDC) declares that a sodium hypochlorite solution of 500 ppm is effective against nearly all bio-contamination. Though many studies support bleach’s disinfectant and sterilant attributes, it does not possess the detergent action for cleaning away soil and residue.
Rotation of disinfecting agents is recommended to prevent the development of resistant microbes. For example, use a 2% bleach solution for 6 days followed by a detergent-based disinfectant on one day. The detergent-based cleaning day may need to be periodically varied to ensure that it is included in the weekly and monthly cleaning routines. Use of a sporicide is highly recommended as part of your monthly or quarterly cleaning cycles. This approach to cleaning solutions is economical as hard-surface disinfectants can cost 15 to 25 times more than bleach solutions.
Consult with manufacturers’ instructions and cleanroom consultants to ensure that your cleaners, disinfectants, and the tools used to apply and wipe them are all compatible. Cleaning agents’ purposes and how they are used vary greatly.
Talk with our cleanroom consultants to ensure your cleanroom wipes and disinfectants are compatible.
Some disinfectants rinsing after the contact time to remove residue. Appropriate rinses in ISO 5 areas include:
• Sterile IPA (70/30 Isopropyl Alcohol/Water)
• Sterile water.
Cleanroom Spills
For spills in the ISO Class 5 area, use a lint-free wipe (nonwoven microfiber wipes offer excellent absorption) to wipe away with sterile water.
Outside the ISO5 area, use a lint-free wipe with tap water.
Then follow with the cleaning agent specified in the SOP to prevent chemical reactions between pharmaceutical components and cleaning agents.
Note that spill prevention is a good practice such as using chemo prep mats to contain spills. Prep mats can save time by avoiding necessary clean-up and reduces risks of compromising your products.

Cleaning Solution Preparation
Following manufacturer instructions is critical to ensure that dilution, storage, applying, and handling of the cleaning agents does not reduce efficacy. The correct dilution ensures that surfaces such as floors, workbenches, and compounding equipment are not damaged as they work to sanitize effectively. Using highly caustic cleaning agents, even used properly, can over time result in pitting stainless steel, especially the stainless steel is not cleanroom-grade.
Material safety data sheets (MSDS) are available from cleaning solution manufacturers and your cleanroom supplier such as Cleanroom Connection. Note as part of USP compliance, MSDS must be kept onsite. Follow manufacturer instructions, keeping these common procedures in mind:
- Mix just before using, documenting the solutions’ preparation in a log.
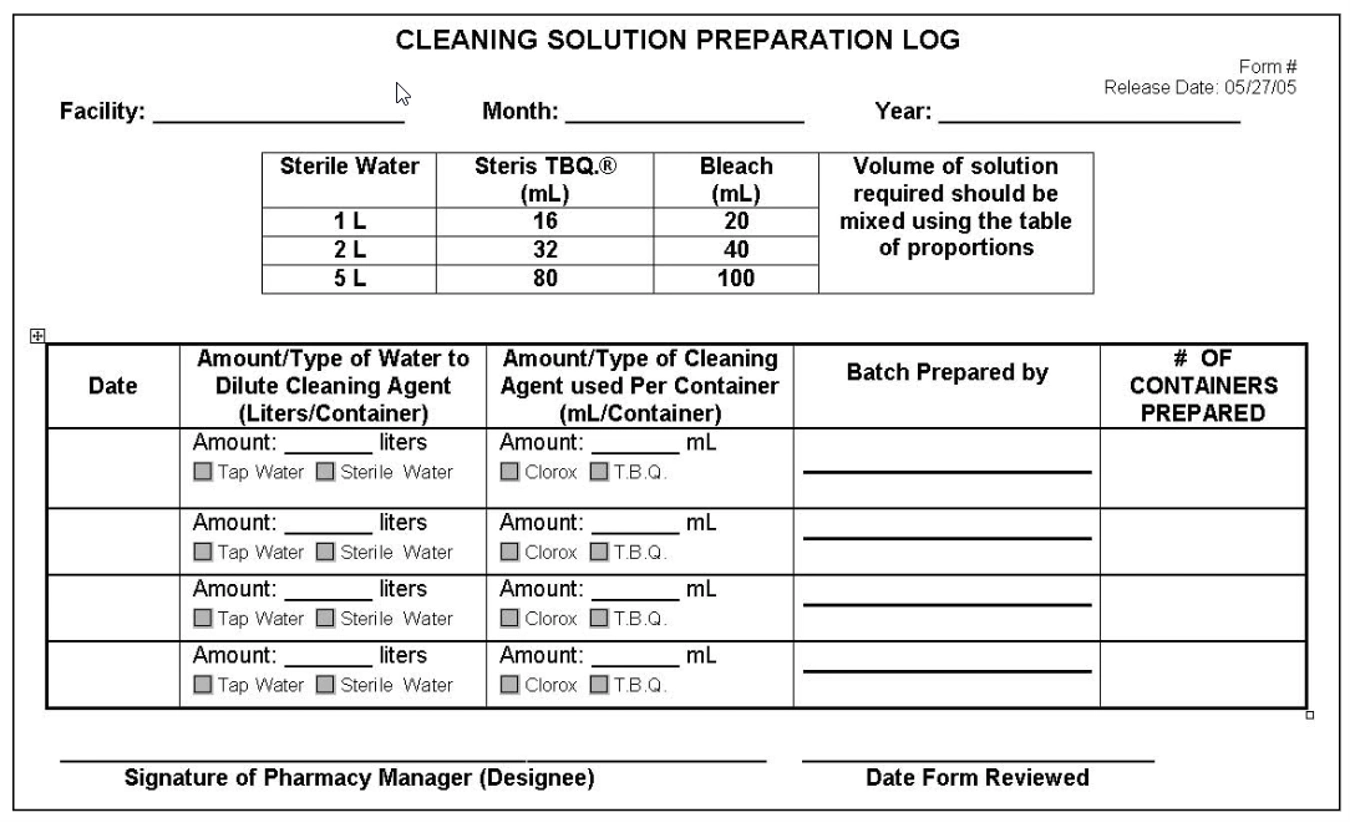
- Pre-measure water into a glass or plastic graduated cylinder or bucket and mix carefully avoiding spillage.
- For ISO Class 5 areas, use sterile water-for-irrigation
- For other areas’ surfaces, tap water may be used.
- Refresh spray-bottle solutions weekly with a new preparation of cleaning solution, then label with expiration date.
Cleaning Equipment and Tools
Mops
Mop handles, heads, and covers as well as buckets must be dedicated to the area in which they are used. Also, to prevent cross-contamination, equipment used to clean floors should not be used to clean walls and ceilings. Equipment used to clean one ISO Class area should not be used to clean different class area.
To reduce the number of mopping “systems” needed for the various cleaning applications, consider moving to higher quality equipment such as autoclavable bucketless cleanroom mop systems and stainless steel cleanroom mop handles appropriate for variety of areas.
Ergonomics is another reason to eliminate buckets in favor of a bucketless mop system as buckets can weigh 18 pounds when filled putting your staff at risk of injury. Cleaning floors, ceilings, and walls using a sprayer filled with cleaning agent in conjunction with a lint-free swivel mop head and cover is effective. Alternatively, use prewetted cleanroom mop heads in lieu of a sprayer. Upfront costs of a bucketless system are higher, but over a short period of time prove less expensive to use. Not only saving on replacement of lower quality equipment, but also fewer staff hours due to reduced pre-mixing of cleaning agents. Plus, your staff will find the system much more user-friendly making following procedures easier.
See our videos on proper sterile mopping technique.
Wipes
Lint-free wipes should be used in ISO Class 5 and higher areas. Larger wipes make covering more surface area faster and easier. Using dry lint-free wipes dipped in the diluted cleaning solution is more economical than pre-wetted wipes to clean surfaces of, for example, laminar airflow hoods, workbenches, furniture, and bio-safety cabinets. But, keep in mind that wipe saturation is important to ensure proper disinfection so in many cases pre-saturated wipes are the most effective and convenient.
The lint-free wipes can also be used to wipe items before bringing them into the critical environment. Be aware that pre-saturated wipes may contain components that cannot be disposed of in unregulated trash, so read the labeling or package insert.
PPE
Cleanroom cleaning staff should be equipped with lint-free, non-particulating apparel:
- Protective clothing:
- For ISO 5, use sterile cleanroom coveralls
- For anteroom (less strict), use non-linting gowns or frocks
- Surgical masks
- Disposable powder-free gloves
- Lint free shoe covers
- Bouffant caps
- Beard covers (if needed)
- Autoclavable safety glasses
PPE should protect the cleaning workers from chemical splash as well as the critical environment from biological and particle contamination.
Cleaning Your Pharmacy Cleanroom
At the beginning of each compounding shift, wipe all ISO Class 5 workstation surfaces including IV bar to the bench surface, interior walls, end panels, and compounders with the cleaning and disinfecting agent specified in the standard operating procedure (SOP). Refer to the daily checklist in our Sample Pharmacy Cleanroom Cleaning Procedure Checklist.
Be mindful to thoroughly wet all the surfaces completely and allow for the appropriate exposure time for the disinfecting solution. Using a “figure 8” pattern to clean or apply solution, moving side to side overlapping the “8’s” creates a more effective cleaning technique. Mops make quick work of flat surfaces especially expansive ones like floor, ceilings, and walls. Lint-free wipers dampened with the cleaning solution are easier to use on smaller and more detailed elements such as molding, handles, and other areas that are not flat. Using a stainless steel or plastic step stool can make cleaning hard-to-reach areas in the compounding room safer and easier.

For USP compliance, maintain a record of cleaning protocols completed. A sample log form is provided to clarify.

While investment in cleaning products and tools is required, the main cost component of cleaning is staff time. The time required to clean and sanitize depend on whether the cleaning routine in process in the daily, weekly, or monthly procedures, the size of the area, the number of workstations, and the experience of the cleaning staff. As a general guide, daily cleaning of a 15’ x 15’ cleanroom with 15’ x 10’ anteroom requires about an hour with weekly cleaning will requiring an additional 20 minutes; monthly cleaning, about two hours.
Cleaning Verification
Your environmental monitoring (EM) program is essential to determining if cleaning is effective. EM is a large topic and mostly outside of this cleanroom cleaning guide, but here are points that help you understand how to verify and where problems may lie if verification shows that cleaning is failing.
When cleaning and disinfecting, you are addressing the removal of both types of particles, viable (micro-organisms) and non-viable (non-living, but provide “transportation” for viable particles). In your USP 797 pharmacy, you must assure asepticism throughout the environment while compounding, testing, or packaging substances that will be consumed by or used topically by humans.
Monitoring for both viable and non-viable particles is important to determining the asepsis of your critical environment.
- Test for bacteria, yeast, and mold by monitoring the air, using contact plates near your staff, and sampling surface areas for any microbial contamination.
- Sample the air for non-viable particles.
In addition to the guidance below, download a detailed brochure of pharmacy cleanroom sampling and verification products.
Monitor Staff
People are the most significant source of contamination in a cleanroom. The first line of defense is not cleaning but rather preventing contamination of both cleanliness and sterility. Your SOP should address specific cleanroom PPE donning procedures to prevent the introduction of viable and non-viable particles.
Using contact plates placed near your staff can help you evaluate how effective your PPE and donning procedures are working. The contact plates monitor for microbial contamination to determine if skin is exposed, PPE is contaminated during donning, or the PPE is not suitable for your environment. After incubation, the contact plates show any bacteria.
Test Cleanroom Surfaces
When monitoring your surfaces include floors, walls, furniture, and equipment. All these areas must be monitored and cleaned appropriately and regularly. Typically, you should monitor regularly for viable particle counts using contact plates that help you detect biological contamination and cross-contamination from previous production.
Contact or witness plates can also be used for surface monitoring by preparing the culture media so that it overflows or sits above the sides of the Petri dish. Then the exposed media is pressed against the surface to capture any contaminants. After incubation, the contact plate will reveal any contamination. Using contact plate applicators can help to standardize this method.
Another method of surface sampling is with sterile swabs. After rubbing the swabs over the area to be sampled, then sub-culturing is used to determine the presence of viable particles the swab picked up. This is the most effective way to sample surfaces that not flat or hard-to-reach.
Sample Air
Depending on the application and level of your facility the HVAC control systems continuously monitor a variety of parameters such as pressure, temperature, and humidity. In addition, monitoring for both viable and non-viable particle counts should be routinely completed. To ensure accuracy of the analysis, air samplers for particle monitoring must be maintained and calibrated regularly.
Air samplers draw in a specific volume of air. Methods used to gather particles in the sampler are:
- Active sampling is when air passes over a media plate that is incubated to reveal viable organisms and indicate the number of particles per cubic volume.
- Settle plates are a passive sampling method that uses Petri dishes that contain sterile growth media (Tryptic Soy Agar). They are left exposed in your facility for up to four hours to allow particles in the air to settle onto it. Then the plate is incubated to reveal contamination. Passive air sampling is less accurate so using a combination of active and passive is recommended.
Increases in non-viable particles should set an investigation into a variety of areas to find the source of particulates. Common issues include improper donning, incorrect product PPE, or incorrect cleaning tools (wipes and mops). If PPE, wipes, and mops that are suitable for less strict facilities are introduced into your ISO5 cleanroom, linting occurs releasing particles. Another source can be products brought into the cleanroom that have not been properly wiped prior to sending through the pass-through.
Increases in viable particles in the air can have similar sources, especially inaccurately followed donning procedures whereby the exterior of the cleanroom apparel is contaminated during donning.
While all these monitoring methods help you determine if and how viable and non-viable particles are introduced, they also help you determine if cleaning protocols are adequately defined and implemented.
Developing Your Cleanroom Cleaning Protocols
Designing a cleanroom cleaning program for a compounding facility requires establishing many policies and procedures and educating staff. It is key to note that this takes time, but pharmacy facilities directors can upgrade the SOP and staff competencies to comply with USP 797’s gowning, handwashing, and cleaning requirements while waiting for other milestones such as approval to build a cleanroom or to implementing secondary engineering controls. Download detailed information about cleanroom classifications.
Explore further by reading about cGMP for your pharmaceutical manufacturing facility.
The investment in facilities and equipment is not prudent if the efficacy of its processes and applications are not maintained correctly. Defining the SOP in rigorous detail regarding procedures, behaviors, practices, equipment, and tools to be employed by your staff is a daunting task. Your cleanroom staff is the key to implementing policies and procedures for effective cleanroom cleaning.
Need help with your cleanroom build or cleanroom cleaning protocols? We can help!
